Biotechnology
China has significantly bolstered its biotechnology sector, prioritizing it in three of the seven cutting-edge science and technology fields in its 14th Five-Year Plan (2021-2025): brain science, genetic engineering, and clinical medicine. Biotech is also crucial to global development goals, particularly in public health, sustainable energy, and food security.
With sizeable domestic support, Chinese companies such as BGI (genome research), Beigene (anti-cancer drugs), Mindray (medical equipment) and WuXi Apptec (services and equipment for drug development) have risen to global prominence.
ChemChina took over Syngenta, the world’s third largest seed producer, in 2017. China is the world’s largest exporter of Active Pharmaceutical Ingredients (APIs).
International collaboration and competition in biotech are intricately intertwined. China participates in roughly a third of the world’s multiregional clinical trials – only the US participates in more. China has also welcomed large investments by multinational pharmaceutical companies in domestic startups. At the same time, fair competition and market access remain a problem for many foreign firms in the healthcare sector, as localization efforts give domestic firms preferential treatment.
Great power competition adds further complexity. China, the US, and Europe all scrutinize the sharing of sensitive genetic and medical data. They also race for supremacy in dual-use technologies, such as brain-computer interfaces, which could be used for military robots.
Graphics dashboard
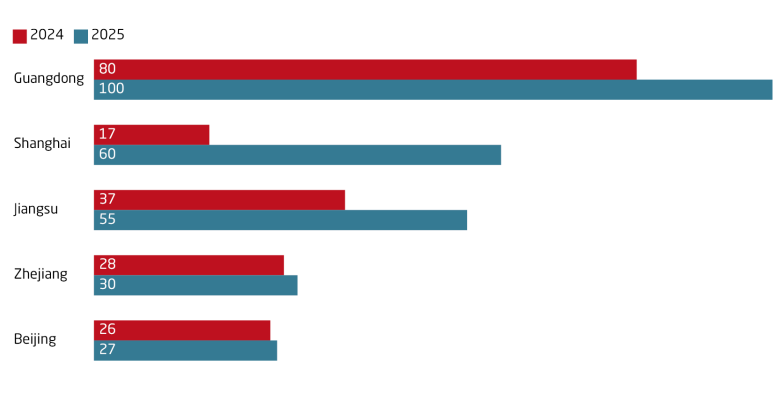
Brain–computer interface (BCI) is an emerging neurotechnology sector in China and one of six “future industries” in the 15th FYP. In March, China approved a BCI device for commercial use, a world first. The device records brain activity and helps people with paralysis regain motor skills like moving and picking up objects. Such approval is a step forward for BCI research. It boosted investor interest, with BCI firms securing EUR 498 million in Q1 2026, more than in all of 2025. There are over 200 BCI firms in China, second only to the US, with major hubs in Guangdong and Shanghai.
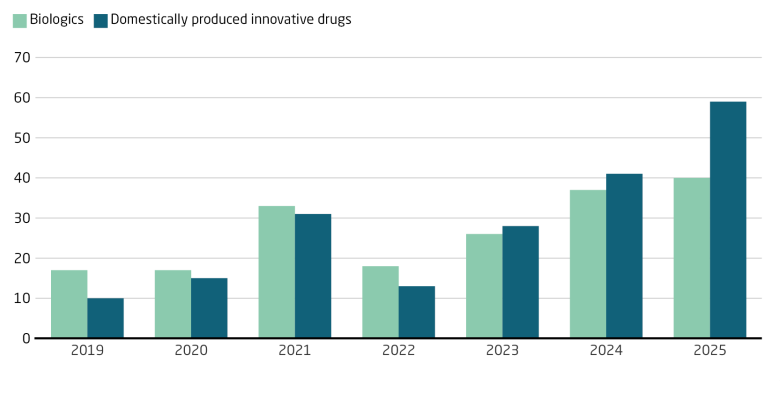
China’s National Medical Products Administration (NMPA) approved 76 innovative drugs for the Chinese market in 2025, which is a 53 percent increase on 2024. The European Medicines Agency (EMA) approved 104 new medicines and 38 new active substances in 2025; a small decline compared to 2024. China’s NMPA attributed its uptick to reforms of the registration review and approval process, adding green lanes for various types of new and urgently needed drugs. However, the rise also reflects a more mature domestic innovation pipeline that caters to growing domestic and international demand for diverse, advanced therapies. Eighty-five percent of NMPA’s innovative drug approvals in 2025 were for applications from Chinese firms. Innovative drugs – treatments with chemical structures or therapeutic uses not yet available on the Chinese market – partly overlap with biologics, which are made from microorganisms, cells, or animal material.
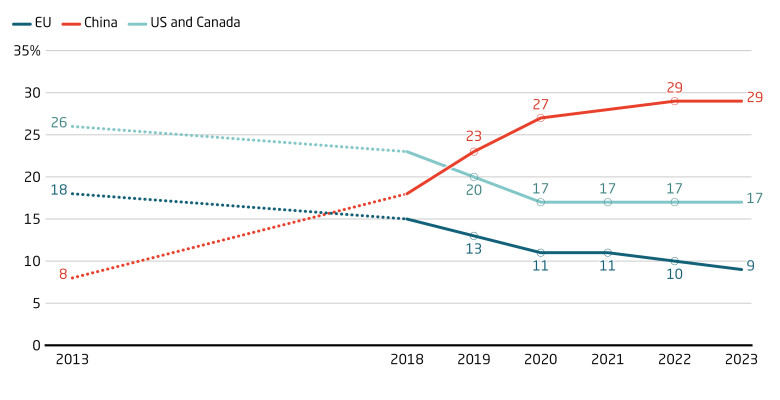
Between 2013 and 2023, China’s slice of global clinical trial starts jumped from 8 percent to 29 percent, overtaking Europe and closing in on North America. Beijing has successfully promoted China as a location for clinical trials. Although getting official approval in China often still takes longer than in Europe, its large public hospitals significantly reduce patient recruitment timelines and costs.
Biotechnology in China: Timeline of crucial events
China approves its first COVID-19 vaccine for general public use, developed by state-backed pharmaceutical giant Sinopharm and the Pfizer/BioNTech vaccine was approved for use in the EU.
Chinese medical device company Mindray Bio-Medical Electronics acquires Finnish biotech company HyTest Invest and its subsidiaries for EUR 532 million.
Leading Chinese medical imaging firm United Imaging invests EUR 410 million in a new manufacturing and research facility in Shanghai to rely heavily on automatic and intelligent processes.
The National Development and Reform Commission releases the 14th Five-Year Plan for the bioeconomy, covering life sciences and biotechnologies and calling for more investment in basic research.
MIIT calls for companies to participate in a new biomedical materials innovation program to increase collaborative innovation with a focus on polymer, metal and inorganic non-metallic materials.
MOST issues clarifying rules on human genetic resources, preventing foreign entities from collecting resources in China and restricting their access to them.
AstraZeneca acquired Gracell Biotechnologies for USD 1.2 billion, marking the first time a multinational drugmaker fully acquires a Chinese biotech firm.
The National Healthcare Security Administration aims to enhance coordination between local governments on centralized or volume-based procurement (high volume, low cost purchase of pharmaceuticals).
Ministry of Science and Technology publishes ethics guidelines on human genome editing. They prohibit clinical research on germline genome editing – i.e. editing in ways that the change is heritable.
Big pharma increases its licensing and acquisition of drugs developed in China, boosting China’s Biotech sector. In 2024, major foreign drugmakers spent USD 3.15 bn on Chinese-developed molecules.
China’s healthcare insurance subsidy increased by 30% to 700 CNY per person, according to the Government Work Report for 2025.
As US-Chinese scientific research further decouples, the US National Institute of Health blocks researchers in China from accessing health databases like the SEER cancer registry.
MIIT unveils a plan for over 20 pilot-scale biomanufacturing platforms, aiming to translate lab research into large-scale industrial production by 2027, serving 200 enterprises.
The brain-computer interface (BCI) industry plans to prioritize healthcare, industrial, and consumer applications until 2030. Its notable achievements include the first human BCI implants.
MIIT and NDRC announce the first batch of 43 companies drafted to build biomanufacturing pilot capacity platforms aiming to pool resources, funding, and talent.
China's first Private Health Insurance Innovative Drug Catalogue includes 19 novel drugs, prioritizing those with “high levels of innovation”, and recommending them for commercial/private healthcare.
The Chinese Academy of Sciences reports progress in brain-computer interface clinical trials, enabling a paralyzed patient to interact in the 3D real world through an embodied intelligence robot.
Chinese pharma companies object to being targeted by the US’s new BIOSECURE Act, which limits federal funding for biotechnology made by companies with ties to “foreign adversaries”.


MIIT issues procurement guidelines, placing restrictions on a wide range of medical equipment, also X-ray machines, MRI & surgical equipment. The rules require 25, 50, 75 or 100 percent local content.
MIIT releases Five-Year Plans for the medical equipment & pharma industries, to ensure basic supplies & improve the industrial chain, enhance pharma innovation, modernization, & supply security 2025.
Chinese healthcare companies such as Andon Health and Beijing Hotgen Biotech see revenue surge thanks primarily to overseas sales of COVID-19 home testing kits.
Fosun Pharma and Genuine Biotech win the right to market the first China-made oral Covid-19 drug. The EU approved the first oral COVID-19 antiviral treatment, Pfizer’s drug Paxlovid, in January 2022.
MIIT releases a three-year plan to develop China’s non-food bio-based materials industry. By 2025, it aims for strong innovation capabilities to broaden its circular economy & reduce carbon emission.
Neusoft Medical launches China's first dual-energy 3.0T magnetic resonance imaging system. Chinese firms can make systems between 1.5T to 3.0T (magnetic field strengths) using only domestic inputs.
Caixin reports: China's outbound deals involving treatment rights exceeded inbound deals for the first time in 2023. Other parties can use a company’s products, technology or intellectual property.
China uses stem cells to develop first open-source brain-computer interface chip to apply in robotics and medical rehabilitation.
China adds 91 items to its list of drugs covered by basic health insurance. It includes innovative drugs, with an average discount of 63%. A growing number – 70% – were from Chinese firms.
China adds the California-based genomic sequencing firm Illumina to its “unreliable entities” list.
Nature publishes findings from the first human transplant of a gene-edited pig liver in a brain-dead recipient - a potential solution to organ shortages -, performed at China’s Xijing Hospital.
MIIT unveils a 2025-2030 pharma digitalization plan. Applications cover AI drug discovery, smart manufacturing, digital clinical trials, and intelligent quality control.
The National Medical Products Administration (NMPA) approves 43 innovative drugs in H1 2025, a year-on-year increase of 59%, highlighting China’s shift toward commercial healthcare.
NMPA approves China’s first brain-computer interface (BCI) medical device standard, effective January 1. The plan details support for core hardware/software and major companies as well as healthcare.
Bayer’s new E-Town Open Innovation Center in Beijing aims to integrate industry, education, research, and entrepreneurship to strengthen China's innovation chain and integration into global markets.
China launches the National Venture Capital Guidance Fund, which features investments for biomedicine and brain–computer interfaces, mainly to attract more private capital and financial institutions.
The EU Commission’s proposal for the European Biotech Act, privileging domestically produced drugs, signals further European decoupling. China may need more factories in Europe for market access.
- US-based Aligos Therapeutics has signed a licensing agreement with China-based Xiamen Amoytop Biotech to develop and commercialize pevifoscorvir sodium for chronic hepatitis B virus (HBV) treatment in China. Under the deal, Aligos will receive an upfront payment of USD 25 million and could earn up to USD 420 million in milestones and royalties. It will retain rights outside of China. (Source (EN): Aligos Therapeutics, April 16, 2026)
- China's National Medical Products Administration (NMPA) released the Implementation Opinion on "AI+drug regulation” to speed up the innovation cycle and integrate AI into the drug regulatory system by 2030. The opinion includes seven priority directions for advancing AI-driven drug oversight and five measures to strengthen digital infrastructure. (Source (CN): NMPA, People.cn, April 2, 2026)
- Swiss drugmaker Novartis will invest over CNY 3.3 billion (USD 460 million) in China. It will upgrade its manufacturing facility in Beijing and strengthen capabilities at its R&D center in Shanghai. (Source (EN/CN): Fiercepharma, Novartis, March 23, 2026)
- British drugmaker AstraZeneca will build a commercial cell therapy manufacturing factory and innovation center in Shanghai. The expansion would make the company the first multinational with end-to-end cell therapy capabilities in China. The facility will produce and supply CAR-T cell therapies, which modify immune cells, for the Asian market. The investment is part of AstraZeneca’s plan to invest USD 15 billion in China through 2030. (Source (EN/CN): Fiercepharma, AstraZeneca, March 19, 2026)
Publications




